Lifecycle Energy Costs of eFuel Production
eFuels can cut lifecycle CO2 but require far more renewable energy and higher costs to produce.
eFuels, or synthetic fuels, are gaining attention as a carbon-neutral energy source. Formula 1 plans to use 100% eFuels by 2026 to support its goal of achieving net zero emissions by 2030. These fuels are made by combining hydrogen (produced via water electrolysis) with CO₂, creating a cleaner alternative to fossil fuels. However, their production is energy-intensive and expensive. Here's what you need to know:
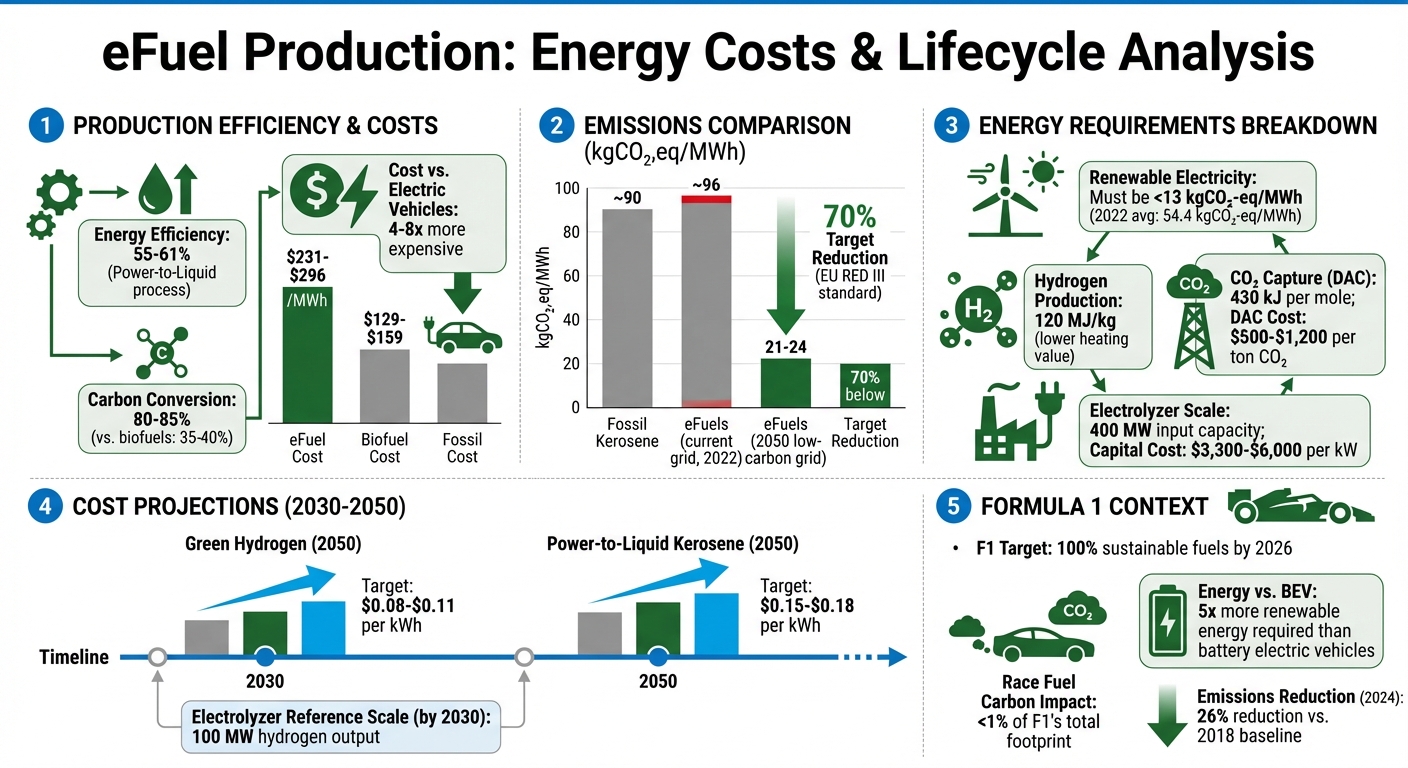
- Energy Demands: eFuel production requires renewable electricity for hydrogen generation, CO₂ extraction, and synthesis processes like Fischer-Tropsch. Current efficiency is around 55–61%.
- Costs: Producing eFuels costs $230–$294 per MWh, significantly higher than fossil fuels and electric vehicle energy.
- CO₂ Reduction: When using low-carbon electricity, eFuels can cut lifecycle emissions by up to 70% compared to traditional fuels.
- Challenges: High costs, reliance on renewable energy, and the need for advanced technologies like Direct Air Capture (DAC) make scaling production difficult.
- Applications: eFuels are ideal for industries where electrification is less practical, such as aviation, shipping, and motorsports.
Formula 1’s adoption of eFuels highlights their potential to decarbonize high-performance engines while leveraging existing fuel infrastructure. However, reducing production costs and improving efficiency are critical for widespread use.
eFuel vs Fossil Fuel Lifecycle Energy Costs and Emissions Comparison
WEBINAR | Critical Next Steps in eFuel Production Development: Reviewing Key Influences on Revenue
Energy Requirements for Raw Material Acquisition
Producing eFuels hinges on three primary components: renewable electricity, hydrogen generated through water electrolysis, and carbon dioxide sourced from the air or industrial processes. Each of these requires a significant energy investment. To genuinely reduce emissions rather than simply move them around, the entire process depends on using low-carbon electricity at every stage.
Renewable Electricity Generation
Renewable electricity forms the backbone of eFuel production. It powers the electrolyzers that split water into hydrogen, the systems that capture CO₂, and the reactors that synthesize liquid fuels. The main hurdle is not just generating enough renewable power but also ensuring a steady supply, given the unpredictable nature of solar and wind energy. To manage these fluctuations, eFuel facilities rely on energy storage solutions like batteries, hydrogen storage tanks, or even underground salt caverns.
The carbon footprint of the electricity used is a critical factor. To meet EU RED III standards, which call for a 70% reduction in greenhouse gas emissions, the electricity must emit less than 13 kg of CO₂-equivalent per MWh. This is far below the 2022 average of 54.4 kg CO₂-eq/MWh. Cleaner electricity can dramatically cut lifecycle emissions, bringing them down from about 96 kg CO₂-eq/MWh to as low as 21–24 kg CO₂-eq/MWh. Additionally, a 30% change in electricity prices can shift eFuel production costs by about 20%, encouraging facilities to set up in areas with abundant, low-cost renewable energy. This renewable power drives the subsequent steps, including water electrolysis and CO₂ capture.
Hydrogen Production Through Water Electrolysis
Water electrolysis, which splits water into hydrogen and oxygen using electricity, is the most energy-demanding part of acquiring raw materials. The sustainability of this "e-hydrogen" depends entirely on the carbon footprint of the electricity used. With a lower heating value of 120 MJ/kg for hydrogen, the energy requirements are substantial.
There are three main electrolyzer technologies:
- Alkaline electrolyzers are the most established and cost-efficient but come with challenges like corrosivity and slower start-up times.
- Proton Exchange Membrane (PEM) electrolyzers are more efficient and better suited for handling fluctuating renewable energy but require pricier electrode materials.
- Solid Oxide Electrolyzer Cells (SOEC) operate at high temperatures, achieving the best efficiency. They can also use waste heat from industrial processes or solar thermal energy, but they need a steady supply of high-temperature heat.
These technologies vary in efficiency and face unique challenges. On top of that, electrolyzers require extremely pure water to avoid electrode damage, which adds to the energy costs for water purification. This step, powered by renewable electricity, sets the stage for capturing CO₂, the next essential input.
CO₂ Capture from Air or Industrial Sources
Once renewable electricity and hydrogen production are in place, the next step is obtaining CO₂. This can come from concentrated industrial sources or directly from the atmosphere. Industrial capture benefits from higher CO₂ concentrations, but integrating these systems into facilities like power plants can increase energy use by 10–40%.
Direct Air Capture (DAC), on the other hand, faces a tougher challenge. Atmospheric CO₂ levels are only about 0.04%, meaning enormous volumes of air must be processed. DAC systems using absorption and electrodialysis typically consume around 430 kJ of energy per mole of CO₂ captured, making them more energy-intensive than industrial methods. However, DAC can be deployed anywhere with renewable electricity, making it possible to produce eFuels even in remote areas far from industrial CO₂ sources.
As of 2022, global CO₂ capture capacity through CCUS stood at 244 million tons annually. But future regulations could shift the landscape. The EU has announced that after 2041, fossil CO₂ emissions from industrial sources will no longer count as "avoided" emissions for renewable fuel production. This shift could make DAC the only viable long-term option, despite its higher energy demands.
Energy Use in Synthesis and Processing
Once raw materials are secured, the next step is converting them into liquid eFuels - a process that requires a significant amount of energy. After obtaining hydrogen and CO₂, the challenge lies in synthesizing them into liquid eFuels. This stage is particularly energy-intensive, especially when it comes to preparing syngas and running the Fischer-Tropsch process. The efficiency of electrolyzers plays a critical role in determining the overall energy consumption of the operation.
Fischer-Tropsch Synthesis
The Fischer-Tropsch (FT) process is used to transform syngas - a mixture of carbon monoxide and hydrogen - into liquid hydrocarbons through polymerization reactions. While the FT reaction itself is exothermic (releases heat), the entire synthesis process consumes a lot of energy. For instance, the Reverse Water Gas Shift (rWGS) reaction, which is used to prepare syngas, operates at a demanding 900°C (1,652°F). Afterward, the Low-Temperature Fischer-Tropsch (LTFT) process runs at temperatures between 356°F and 482°F (180–250°C) to produce the desired hydrocarbon chains.
To optimize the output, long-chain paraffins (also known as FT waxes) are subjected to hydrocracking. This additional catalytic step uses hydrogen to break down the chains into middle distillates, such as jet fuel. Maintaining an optimal hydrogen-to-carbon ratio is crucial for efficiency and is managed through adjustments in the rWGS reaction or electrolyzer tuning. The carbon conversion efficiency for eFuels using FT synthesis typically ranges from 77% to 85%, while the overall energy efficiency for Power-to-Liquid (PtL) processes falls between 55% and 61%. As Diego Freire Ordóñez and colleagues pointed out:
"The energy-intensive nature, high production costs and the need for green H₂ and concentrated CO₂ streams constitute the main barriers to widespread deployment".
These energy demands significantly influence the lifecycle energy costs of eFuels. However, advancements in process efficiencies - such as scaling electrolyzer operations - can help mitigate these challenges.
Electrolyzer Scale and Efficiency
The performance of electrolyzers is a key factor in reducing the energy intensity of the synthesis process. The scale and efficiency of these devices have a direct impact on the energy consumed during production. Industrial-scale eFuel plants typically operate at an input capacity of 400 MW to achieve economies of scale, though the capital costs remain high, ranging from $3,300 to $6,000 per kW of output. Syngas production alone, which includes electrolysis, accounts for 50%–68% of the total investment required for such facilities.
Among available technologies, Solid Oxide Electrolyzer Cells (SOEC) are the most efficient, especially when paired with industrial waste heat or solar thermal sources. This integration reduces the external electrical demand for water splitting and aligns with the high-temperature needs of syngas production. Proton Exchange Membrane (PEM) electrolyzers also offer high efficiency and are better suited for handling the variable energy output of renewable sources compared to Alkaline Electrolyzer Cells (AEC). Additionally, hybridizing energy sources with e-biofuels can further improve efficiency, reaching levels of approximately 61.3% to 64% by optimizing the carbon-to-hydrogen balance. Scaling up and fine-tuning electrolyzer performance is essential for minimizing the overall energy footprint of eFuel production.
Energy Costs in Distribution, Storage, and End-Use
After the energy-intensive synthesis phase, eFuels benefit from their inherent stability, which helps maintain an efficient energy profile during distribution, storage, and combustion. These fuels integrate seamlessly into existing hydrocarbon infrastructure, as highlighted by Corinne Stollery, Consultant at io consulting:
"The pre-existence of hydrocarbon infrastructure means that efuels can be directly and instantly integrated into the current fossil fuel supply chain and propulsion systems, allowing for a fast and simple transition solution."
Transportation and Storage Energy Losses
Transporting and storing eFuels comes with minimal energy losses compared to their hydrogen precursors. Once hydrogen and CO₂ are converted into liquid eFuels like e-diesel or e-kerosene, they remain stable at ambient temperatures and pressures. This stability allows them to be transported using existing pipelines, tankers, and refueling stations without requiring any modifications. By co-locating renewable energy production, electrolysis, and carbon capture at the synthesis site, energy losses are further reduced, as the energy-dense liquid fuel becomes the primary product transported - not gaseous hydrogen. This streamlined process ensures efficient distribution, paving the way for effective combustion.
Combustion Energy Output and Efficiency
When combusted, eFuels deliver energy outputs similar to traditional fossil fuels. The Fischer-Tropsch synthesis process creates high-purity n-alkanes (paraffins) that are free of sulfur and aromatics, resulting in cleaner combustion compared to crude-derived fuels. This chemical purity is especially advantageous in high-performance settings like Formula One, where fuel quality directly influences engine performance and reliability.
Although eFuels face the same thermodynamic constraints as fossil fuels and still produce NOx and particulate emissions that require management, their gravimetric energy density far surpasses that of batteries. This makes them particularly suitable for applications like aviation and motorsports. Jeffrey Goldmeer, Senior Director of Technology Strategy at GE Vernova, emphasizes this point:
"e-fuels have the potential to positively affect our carbon footprint without changing how we power a large portion of the transportation sector including trucking, rail, aviation, and marine shipping."
The efficiency lies in the ability to continue using high-performance engines without redesigning them, while the upstream use of renewable electricity and carbon capture addresses the lifecycle carbon footprint.
Cost Projections and Efficiency Improvements
The economics of eFuel production are set to change significantly over the next two decades as advancements in technology and decreasing renewable energy costs reshape the landscape. These shifts are critical because they directly influence the overall energy footprint of eFuel production throughout its lifecycle.
Energy Cost Projections for 2030-2050
By 2050, the median levelized costs are projected to drop to $0.08–$0.11 per kWh for green hydrogen and $0.15–$0.18 per kWh for power-to-liquid kerosene. These reductions are driven by factors like improved learning curves, economies of scale, and a cleaner power grid.
The pace of these cost reductions will depend on the global climate trajectory. Under a "business-as-usual" scenario, key technologies like high-temperature steam electrolysis may not reach full commercial maturity until 2050. However, under more ambitious 1.5°C climate pathways, these technologies could achieve this milestone by 2030, accelerating cost declines. Electrolyzer technologies - such as Proton Exchange Membrane (PEM), Alkaline Electrolysis (AE), and Solid Oxide Electrolysis Cells (SOEC) - are expected to hit a reference scale of 100 MW of hydrogen output by 2030.
Financing plays a pivotal role in these advances. Capital costs often dictate project viability, with the Weighted Average Cost of Capital (WACC) being a key factor. This is particularly true in areas rich in renewable resources but perceived as higher risk. As researchers emphasize:
"Technology innovation must go hand-in-hand with investment de-risking and international policy coordination to enable a resilient, equitable, and cost-effective global energy transition".
These shifting cost dynamics highlight the importance of strategic site selection to optimize production economics.
Reducing Costs Through Site Selection
Geography and infrastructure are just as critical as technological advancements in achieving cost efficiency. Regions like North Africa and the Iberian Peninsula, with their abundant renewable resources and favorable investment conditions, are emerging as top contenders for low-cost eFuel production. These areas benefit from high solar and wind capacity factors, reducing the need for extensive energy storage and improving plant utilization.
To further cut costs, facilities must carefully balance solar and wind inputs to avoid renewable curtailment - when surplus energy is discarded because it exceeds immediate demand. However, the variability and seasonality of renewable energy require significant investments in electricity, hydrogen, and CO₂ storage to ensure uninterrupted plant operations. For example, a 30% fluctuation in electricity prices can lead to a 14% cost variation for e-biofuels and a 20% change for pure e-fuels. This makes access to affordable renewable power a cornerstone of reducing lifecycle energy costs.
Integrating eFuel facilities with existing oil refineries offers additional economic benefits, such as shared infrastructure and reduced capital requirements. However, the decision between domestic production and importing eFuels from low-cost regions must account for transportation and storage energy losses. Fortunately, these losses are minimal for liquid eFuels compared to their hydrogen precursors, making them a more efficient option for international trade.
Formula One Applications and Fuel Comparisons
Formula One's push toward 100% sustainable fuels by 2026 and carbon neutrality by 2030 places eFuels at the forefront of its environmental strategy. While race fuel contributes less than 1% of the sport's total carbon footprint, this shift has a broader aim: demonstrating that high-performance internal combustion engines can operate sustainably without losing the power, sound, or quick refueling that define F1. This effort not only tackles production challenges but also introduces new performance and sustainability dynamics to the sport.
eFuels vs. Fossil Fuels: Lifecycle Energy Comparison
The energy lifecycle of eFuels shows a trade-off: higher production energy leads to operational emissions benefits. eFuels convert carbon at rates of 80–85%, far outperforming the 35–40% seen in standard biofuels, though their production demands significant energy. In fact, producing synthetic fuels requires five times more renewable energy than battery electric vehicles.
The financial aspect is another hurdle. Producing eFuels costs between $231–$296 per MWh - far higher than biofuels, which cost $129–$159 per MWh. Additionally, eFuels are 4 to 8 times more expensive than operating electric vehicles. A large part of this cost comes from Direct Air Capture (DAC) technology, used to extract CO₂ from the atmosphere, which costs $500 to $1,200 per ton.
Despite these challenges, eFuels bring distinct advantages for motorsport. They retain the high energy density and rapid refueling capabilities essential for F1 race strategies, while theoretically adding no new carbon to the atmosphere. The 2026 F1 fuel is being developed as a "drop-in" solution, meaning it will work seamlessly with current hybrid power units that combine internal combustion engines with 350 kW electric motor-generators.
However, performance remains a key concern. As Toto Wolff highlighted, the green supply chain significantly increases costs. Sustainable eFuels generally have a lower energy density than traditional gasoline, and any reduction in combustion stability could negatively impact performance under F1's strict energy limits.
eFuels and Formula One's Carbon Neutrality Goals
Beyond energy and cost considerations, eFuels are central to F1's larger decarbonization goals. With 1.4 billion internal combustion engine vehicles still on the roads globally, F1 and the FIA's SRFAS initiative - which mandates at least 99% sustainable components - position the sport as a high-profile testing platform for reducing emissions in existing transportation systems without requiring mass vehicle replacement.
F1 has already made strides outside of race fuel. By the end of 2024, the sport achieved a 26% reduction in total carbon emissions compared to 2018, largely by improving logistics, which account for two-thirds of its carbon footprint. For example, F1's European truck fleet now uses biofuels, cutting emissions by an average of 83% compared to traditional diesel.
Fraser Browning, founder of Kurb Carbon, underscored the ripple effect of motorsport innovation:
"I believe that we wouldn't have hybrid vehicles in the form and as quickly as we do today if it hadn't been for input from sporting innovation".
While critics argue that battery-electric vehicles are more energy-efficient for passenger transport, eFuels offer a solution for sectors like motorsport, aviation, and heavy freight, where high energy density and rapid refueling are critical.
Scaling production while maintaining F1's performance standards remains a significant challenge. However, the lifecycle improvements mentioned earlier provide a technical roadmap for achieving F1's sustainability goals. Under a 2050 low-carbon electricity mix, eFuel emissions could drop to 21–24 kgCO₂,eq/MWh - well below the approximately 90 kgCO₂,eq/MWh of fossil kerosene. To meet EU RED III sustainability standards, eFuels must achieve 65% to 70% lower greenhouse gas emissions over their lifecycle compared to fossil fuels - an ambitious target that depends on a largely decarbonized electricity grid.
Conclusion
eFuels achieve a carbon conversion rate of 80–85%, significantly outperforming traditional biofuels, which convert just 35–40%. However, their production costs remain steep, ranging from $231 to $296 per MWh. This is largely due to the electricity required and its carbon intensity. These figures offer a clear perspective on how eFuels align with Formula 1's sustainability goals.
The energy density and rapid refueling capabilities of eFuels make them an ideal match for motorsport's unique demands. When produced using a decarbonized electricity grid, their emissions can be remarkably low. For example, under a projected 2050 low-carbon energy mix (13 kgCO₂,eq per MWh), eFuel emissions could fall to 21–24 kgCO₂,eq per MWh, far below the approximately 90 kgCO₂,eq per MWh of fossil kerosene.
Scaling up electrolyzer technology and strategically locating production facilities near renewable energy sources are essential steps forward. As Etienne de Chambost explained:
"The 'defossilization' of carbonaceous molecule production also needs alternative energy sources".
While battery-electric systems dominate passenger transport, eFuels are indispensable in sectors requiring high energy density, such as motorsport, aviation, and heavy freight. These industries benefit from the rapid refueling and performance characteristics highlighted in this analysis. Formula 1's pledge to transition to 100% sustainable fuels by 2026 positions it as a key innovator in demonstrating how eFuels can transform internal combustion engines worldwide.
This examination underscores the potential of refining eFuel production to balance high performance with environmental responsibility. Although current costs are high, the path forward is clear: investing in renewable energy infrastructure and improving electrolyzer efficiency. For Formula 1 and beyond, eFuels represent more than just a racing solution - they are a vital step toward proving that high-performance combustion can thrive alongside a sustainable future. The continued optimization of production processes and renewable energy integration will be crucial in achieving this vision.
FAQs
Why do eFuels need so much electricity?
eFuels need a lot of electricity due to the energy-heavy steps involved in their production. For instance, water electrolysis, which is used to produce hydrogen, operates at an efficiency of about 70–80%. Beyond that, turning hydrogen into eFuels through processes like Fischer–Tropsch synthesis also consumes considerable energy. To keep the carbon footprint low, renewable energy plays a critical role. This means having access to plentiful and affordable electricity is essential to making eFuels both environmentally friendly and economically viable.
Are eFuels actually carbon-neutral over their lifecycle?
E-fuels, while offering some environmental benefits, are not entirely carbon-neutral throughout their lifecycle. Producing them requires a significant amount of energy, making the process more expensive than using traditional fossil fuels. Additionally, their production can have environmental consequences, potentially affecting both human health and ecosystems. That said, when e-fuels are created using renewable energy sources and direct air capture technology, they can help reduce overall CO₂ emissions.
What would make eFuels cheaper to scale up?
Reducing the cost of eFuels means tackling the expenses tied to raw materials, energy use, and the conversion processes involved. Key approaches include boosting production efficiency, integrating advanced technologies, and scaling up operations to take advantage of economies of scale. Over time, costs can also drop with the help of supportive policies, such as tax incentives or carbon pricing. Creating stronger market demand and cutting dependence on fossil fuels can further drive down consumer prices, making eFuels more accessible and encouraging wider use.




